Japan’s R&D and Application Practices toward the Realization of induced Pluripotent Stem Cell (iPS Cell) Therapy
Topics
Promotion of Research and Development on iPS Cells
Induced pluripotent stem (iPS) cells, or iPSCs, were first artificially created in 2006 by the research group led by Professor YAMANAKA Shinya of Kyoto University. iPSCs have the potential to differentiate into any type of cell, such as nerve cells, muscle cells, and organ cells. The application of iPSCs in medicine is expected to contribute to the elucidation of disease causes, improved disease modeling, drug screening and the development of regenerative medicine products. As is well known, Professor Yamanaka was awarded the 2012 Nobel Prize in Physiology or Medicine for this achievement.
Since its establishment in 2015, AMED has continuously supported medical research and development using iPSCs for regenerative medicine, drug discovery research, disease modeling, and basic and fundamental research. These efforts have been carried out in collaboration with the Cabinet Office, the Ministry of Education, Culture, Sports, Science and Technology (MEXT), the Ministry of Health, Labour and Welfare (MHLW), and the Ministry of Economy, Trade and Industry (METI), under the government of Japan’s “Health and Medical Strategy”.
Approval of iPS Cell-Based Regenerative Medicine Products
On March 6, 2026, MHLW granted approval for the marketing of two iPS cell-based regenerative medicine products; “RiHEART®” and “AMCHEPRY®” with conditions and time limits. These official approvals mark the world's first practical application of iPS cell-based therapeutic products.
“RiHEART®” is an iPS cell-derived cardiomyocyte transplantation therapy for ischemic cardiomyopathy. This product was developed and applied by Cuorips Inc., based on the R&D achievements of Prof. SAWA Yoshiki’s research group at the Graduate School of Medicine at the University of Osaka. “AMCHEPRY®” is an iPS cell-derived neural progenitor cell transplantation therapy for Parkinson’s disease. This product was developed and applied by Sumitomo Pharma and is based on the R&D achievements of Prof. TAKAHASHI Jun’s research group at the Center for iPS Cell Research and Application at Kyoto University.
Japan’s conditional and time-limited approval scheme for regenerative medicine products requires confirmation of safety and a reasonable prediction of efficacy based on the results of clinical trials. This scheme is premised on the understanding that the products’ efficacy will be essentially confirmed and their safety further evaluated after the marketing, followed by re-application for full approval within the designated time-period.
AMED’s Support for Medical Research and Development using iPSCs
AMED supports R&D of new medical technologies using iPSCs, as well as their practical application. Prior to the establishment of AMED, the government of Japan and related public organizations provided support for basic and fundamental research on iPSCs, and their practical applications, through programs such as the Grants-in-Aid for Scientific Research, the Strategic Basic Research Program (CREST) of the Japan Science and Technology Agency (JST), and the Health and Labor Sciences Research Grants. With the understanding and cooperation of many stakeholders, including researchers, medical doctors, policymakers, business leaders, and patients, the delivery of iPS cell-based regenerative medicine products to society this year represents a significant milestone, marking the 20th anniversary since the initial establishment of iPSCs.
As Japan’s public research funder in healthcare and medical fields, AMED has been providing nationwide support for R&D projects on a wide range of iPS cell-based therapeutic products and technologies through collaborative initiatives involving MEXT, MHLW, and METI.
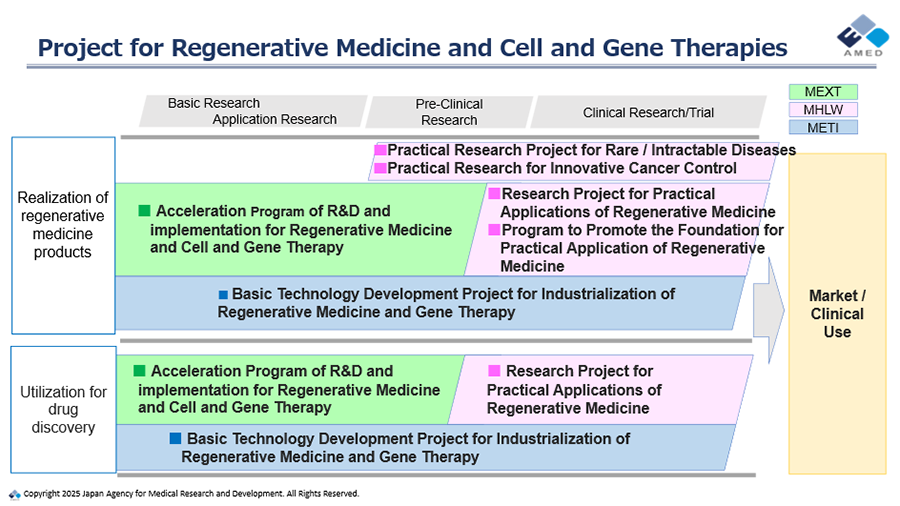
AMED will continue to proactively advance essential projects to deliver outcomes to patients and their families as quickly as possible, through the promotion of medical R&D and enhancing the supporting environment, including support for the creation of new iPS cell-based medical technology seeds and their practical application.
Note: Please refer to the Japanese original for any references to institutional procedures, or official project names etc.
Links
- Project for Regenerative Medicine and Cell and Gene Therapies
- Graduate School of Medicine/ Faculty of Medicine, the University of Osaka
- Cuorips Inc.
- Center for iPS Cell Research and Application Kyoto University
- Sumitomo Pharma
- PMDA [Pharmaceuticals and Medical Device Agency] - Regenerative Medical Products -
03/23/26
Last updated 03/23/26

