Dysbiosis in the Gut Microbiome Shapes Disease Course in the Different Stages of Multiple Sclerosis - Combination of microbial composition and comprehensive functional gene analyses revealed the key microbial function associated with secondary progressive multiple sclerosis -
News Releases & Research Results
National Center of Neurology and Psychiatry (NCNP)
RIKEN
Japan Agency for Medical Research and Development (AMED)
Collaborative research team by Takashi Yamamura (director of the Multiple Sclerosis Center, National Center of Neurology and Psychiatry), Wataru Suda (deputy team leader of the Laboratory for Microbiome Science, RIKEN), and Masahira Hattori (professor of the Waseda University) revealed the ecological and functional differences in the gut microbiome between patients with relapsing-remitting multiple sclerosis (MS) and secondary progressive MS.
In the gut of secondary progressive MS, an enhancement in oxidative stress and microbial DNA repair was observed, which suggested the possible association between gut-derived oxidative stress and chronic neuroinflammation. These are breakthrough results leading to the innovative treatment for the progressive stage of MS.
These results are published in Proceedings of the National Academy of Sciences of the United States of America (PNAS) at August 24, 2020 at 3:00 PM U.S. Eastern time.
This study was supported by the Practical Research Project for Rare/Intractable Diseases from Japan Agency for Medical Research and Development (AMED) and AMED-CREST.
Background
Multiple sclerosis (MS) is an autoimmune demyelinating disease of the central nervous system (CNS), which is the major neurological disease with onset in young adulthood. The number of patients with MS has been increasing for decades in developed countries, but definitive treatment has not been established. Most patients experience signs of relapse with intervals of remission lasting for months to years during their early clinical stage. Patients who are staying in this stage are referred to as relapsing-remitting MS. Proportions of patients with relapsing-remitting MS may shift to the progressive disease form referred to as secondary progressive MS in years after onset of MS. Once transition to secondary progressive MS is established, neurological disability such as gait difficulty accumulates gradually.
A typical MS is a distinct disease condition, in which spinal cord and optic nerves are mainly affected, but only few brain lesions are detected by MRI scans. Neuromyelitis optica spectrum disorder (NMOSD) was previously regarded as a subtype of MS, but is now considered an independent disease, since the majority of the cases are characterized by the presence of anti-aquaporin 4 autoantibody.
Even though the treatment for MS has dramatically changed after entering the 21st century, there is almost no effective drug for secondary progressive MS. The development of innovative therapy for secondary progressive MS is the biggest “unmet medical need” in this field.
Yamamura et al. previously reported the effects of oral antibiotics on CNS inflammation in an experiment using a mouse model of MS in 2008 (*1). This study was the beginning and urged many researchers to analyze the association between gut microbiome and CNS inflammation. Moreover, Yamamura et al. also reported the dysbiosis in the gut microbiome of patients with relapsing-remitting MS in 2015 (*2). Works from the research groups all over the world reported the results regarding the gut microbiome in patients with relapsing-remitting MS. All of these findings strengthened the association between MS and gut microbiome.
As the next step, many researchers have great interests in the gut microbiome of patients with secondary progressive MS. We conducted this research to reveal the characteristics of gut microbiomes in patients with secondary progressive MS by analyzing the microbial composition and functional data based on 16S ribosomal RNA (16SrRNA) gene and metagenomic analyses.
Research content
Microbial DNA was isolated and purified from the fecal samples of 62 patients with relapsing-remitting MS, 15 patients with secondary progressive MS, 21 patients with atypical MS, and 20 patients with NMOSD in National Center of Neurology and Psychiatry hospital. We analyzed the microbial DNA using a next-generation sequencer based on metagenomic analysis. Sequence data from 55 healthy Japanese subjects were also included in the present study. We then compared the microbial composition and functional gene data and obtained the following results.
1) Microbial composition analysis
We revealed the microbial species increased or decreased in the gut of various subtypes of MS compared with healthy Japanese subjects. Moreover, we distinguished the total patients with each MS subtype into high- and low-severity groups based on clinical information including disease activity and neurological disability, and revealed the specific species strongly associated with disease severity in each MS subtype (Figure 1).
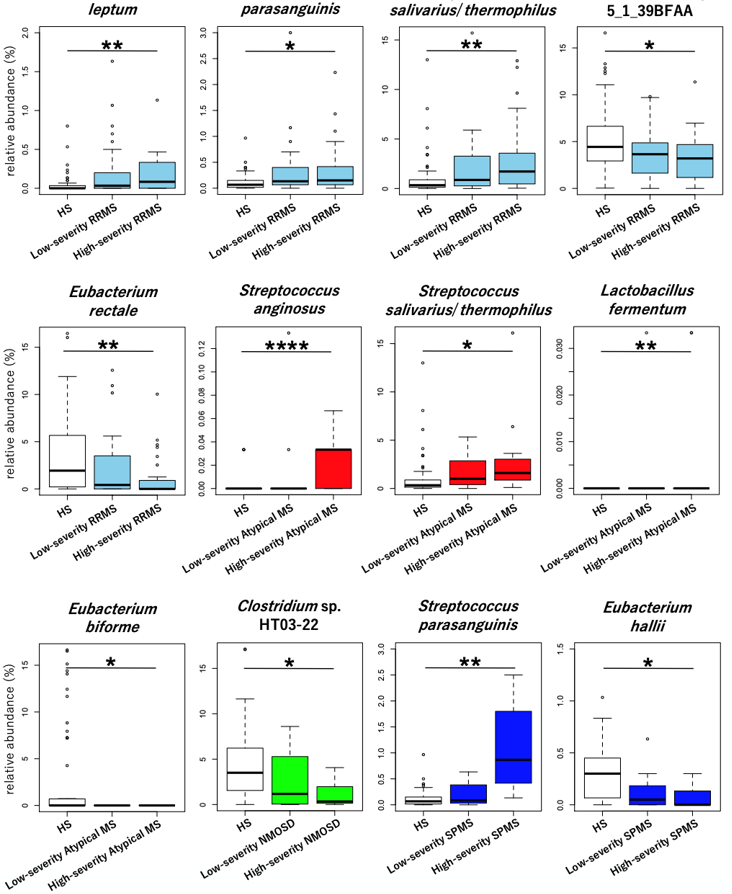
2)Functional analysis of the gut microbiome
We analyzed gene information of the entire gut flora based on the metagenomic analysis to reveal the microbial composition and function of the gut microbiome. Gene functions and metabolic pathways including those genes detected from the obtained sequence data were analyzed using the Kyoto Encyclopedia of Genes and Genomes (KEGG) database. In the present study, we particularly focused on the comparisons between healthy subjects and relapsing-remitting MS patients and between relapsing-remitting MS patients and secondary progressive MS patients to reveal the microbial function associated with the development and progression of MS.
2-1)Comparison between healthy subjects and relapsing-remitting MS patients
6,163 genes regarding the gut microbial function were detected in the present study. Among them, 97 genes were increased and 117 were decreased in relapsing-remitting MS patients compared with healthy subjects. Next, we ranked the metabolic pathways including these genes in a descending order of the degree of increase or decrease between the two groups by utilizing a statistical approach. Based on this ranking, the ability of gut microbiome to produce butyrate and propionate was clearly depleted in relapsing-remitting MS patients compared with healthy subjects (Figure 2).
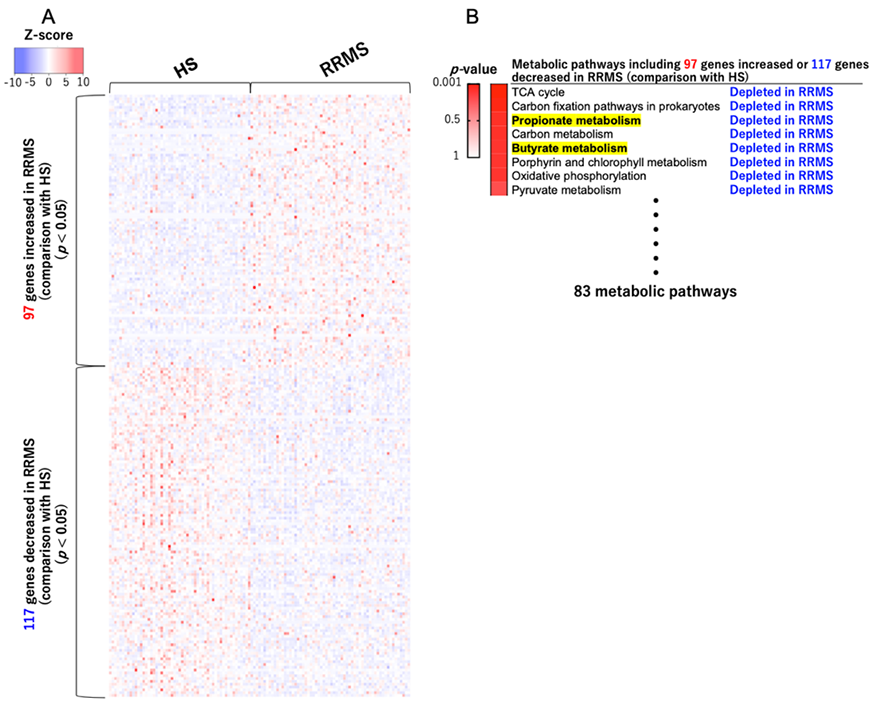
(A) 97 genes were increased and 117 were decreased in patients with relapsing-remitting MS compared with healthy subjects. (B) We ranked the metabolic pathways including these genes in a descending order of the degree of increase or decrease between the two groups. RRMS; relapsing-remitting MS, HS; healthy Japanese subjects
2-2)Comparison between relapsing-remitting MS patients and secondary progressive MS patients
In the comparison between relapsing-remitting MS patients and secondary progressive MS patients, 38 genes were increased and 14 were decreased in secondary progressive MS patients compared with relapsing-remitting MS patients. We ranked the metabolic pathways including these genes in a descending order of the degree of increase or decrease between the two groups. Based on this ranking, the mismatch repair pathway associated with DNA damage was enhanced in secondary progressive MS patients compared to relapsing-remitting MS patients (Figure 3).
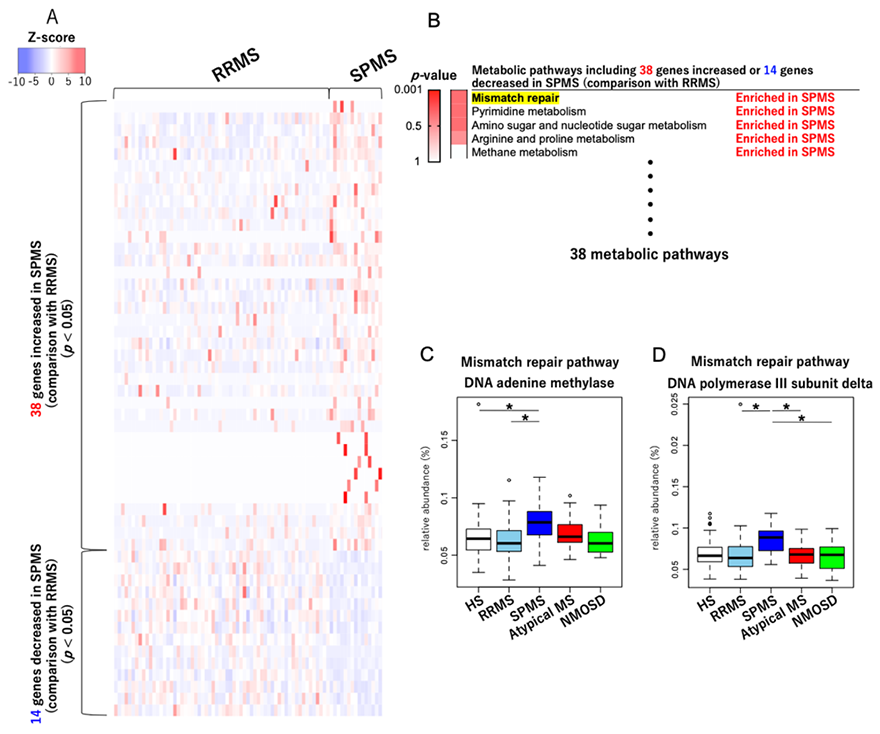
(A) 38 genes were increased and 14 were decreased in patients with secondary progressive MS compared with healthy subjects. (B) We ranked the metabolic pathways including these genes in a descending order of the degree of increase or decrease between the two groups. (C, D) Among genes included in the mismatch repair pathway, the relative abundances of two genes were increased in secondary progressive MS patients compared to relapsing-remitting MS patients. RRMS; relapsing-remitting MS, SPMS; secondary progressive MS, *p <0.05 (Wilcoxon rank sum test)
3)Fecal metabolite analysis
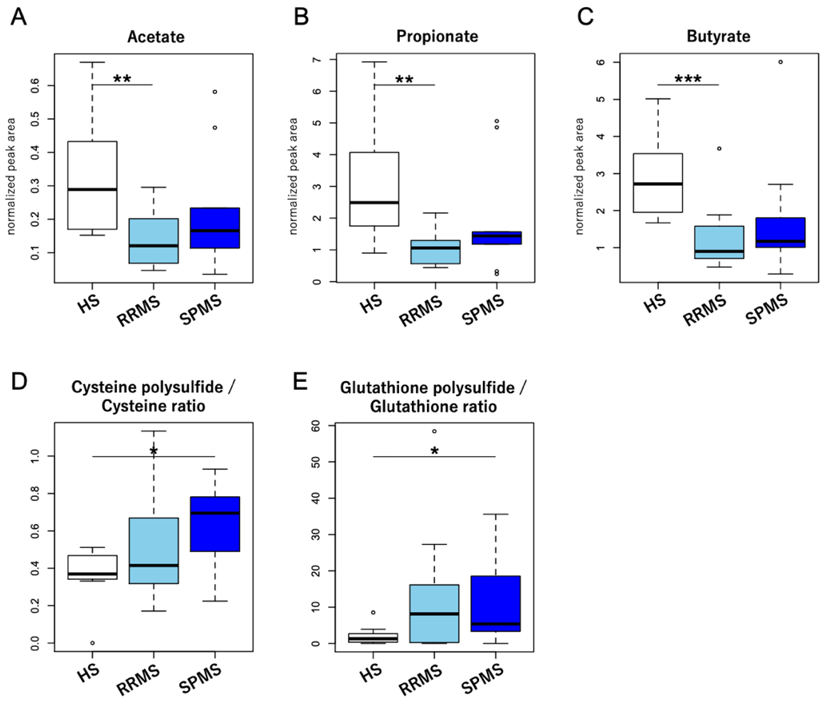
Finally, to confirm the obtained results from the microbial functional analysis, we analyzed fecal metabolites obtained from 12 relapsing-remitting MS patients, 9 secondary progressive MS patients, and 8 healthy subjects. As expected, the concentration of butyrate and propionate was markedly decreased in the feces of patients with relapsing-remitting MS compared with that of healthy subjects.
We also conducted sulfur metabolomics to quantify the degree of fecal oxidation. The degree of polysulfidation in sulfur compounds reflects the degree of oxidative stress in the surrounding environment (*3). The degree of polysulfidation in sulfur compounds such as cysteine and glutathione was increased in the feces of patients with secondary progressive MS (Figure 4), which suggested the enhanced oxidative stress in the gut of secondary progressive MS.
Discussion
Gut microbiome-derived butyrate and propionate could not only induce colonic regulatory T cells but enhance remyelination in the CNS. Dysbiosis in the gut microbiome and reduction of butyrate and propionate in the gut are at least in part, associated with the exacerbation of relapsing-remitting MS.
On the other hand, enhanced oxidative stress in the gut of patients with secondary progressive MS might provoke microbial DNA damage and subsequently enhanced mismatch repair. These findings suggested that gut-derived oxidative stress is closely associated with chronic neuroinflammation in the pathogenesis of secondary progressive MS.
Significance of research
We revealed the characteristics of gut microbiome in each of relapsing-remitting MS and secondary progressive MS (Figure 5). This is a fruitful achievement leading to the development of innovative therapy for secondary progressive MS. Furthermore, healthy Japanese gut microbiome is characterized by the abundant short chain fatty acid production and low microbial DNA damage (*4), which may have implications for understanding why prevalence of MS is relatively smaller in Japan, as compared with western countries. The present study emphasizes the importance of microbiome study from Japan among various efforts aimed at overcoming MS. The era of radical cure for MS by controlling the gut microbiome will come in the near future.
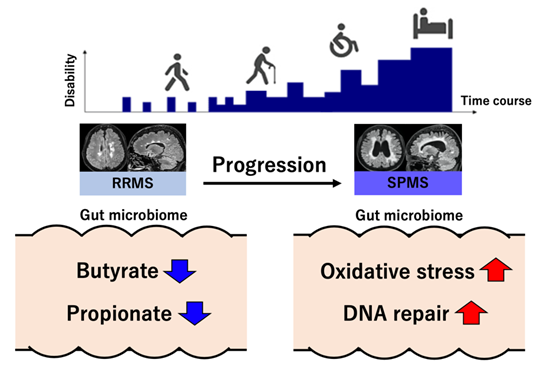
RRMS; relapsing-remitting MS, SPMS; secondary progressive MS
References
*2 S. Miyake et al., Dysbiosis in the Gut Microbiota of Patients with Multiple Sclerosis, with a Striking Depletion of Species Belonging to Clostridia XIVa and IV Clusters. PLoS One 10, e0137429 (2015).
*3 K. Yamada et al., Characterization of sulfur-compound metabolism underlying wax-ester fermentation in Euglena gracilis. Scientific reports 9, 853 (2019).
*4 S. Nishijima et al., The gut microbiome of healthy Japanese and its microbial and functional uniqueness. DNA research 23, 125-133 (2016).
Article information
- Title
- “Alterations of the gut ecological and functional microenvironment in different stages of multiple sclerosis”
- Authors
- Daiki Takewaki, Wataru Suda, Wakiro Sato, Lena Takayasu, Naveen Kumar, Kimitoshi Kimura, Naoko Kaga, Toshiki Mizuno, Sachiko Miyake, Masahira Hattori, and Takashi Yamamura
- Article
- Proceedings of the National Academy of Sciences of the United States of America
- DOI:
- 10.1073/pnas.2011703117
Contact
Media Inquiries
National Center of Neurology and Psychiatry.
Takashi Yamamura MD,PhD
Director and Chief of the Neuroimmunology Section
Tel: +81-42-341-2711
Email: yamamura(at)ncnp.go.jp
RIKEN
Email: ex-press(at)riken.jp
Japan Agency For Medical Research and Development (AMED)
Practical Research Project for Rare / Intractable Diseases
Email: nambyo-r(at)amed.go.jp
Advanced Research & Development Programs for Medical Innovation
Email: kenkyuk-ask(at)amed.go.jp
* Replace "(at)" with "@".
Links
08/25/20
Last updated 08/25/20

