Advanced Research & Development Programs for Medical Innovation
Outline
| Key Fields | Basic Research for Medical Innovation |
|---|---|
| R&D phase | Basic Study |
| Contact |
|
Overview
This project develops time-limited research frameworks across organizational boundaries and promotes cutting-edge research and development designed to create and incubate revolutionary breakthroughs with the goal of creating innovative drugs, medical devices, medical technologies, and other innovations, as well as ensuring that research projects on promising results proceed more rapidly and in greater depth.
Program Outline
With the goal of developing innovative drugs, medical devices, and medical technologies under R&D objectives determined by the government, researchers in universities and other institutions are invited to submit R&D proposals upon which a time-limited R&D system transcending organizational frameworks for driving R&D activities will be constructed. The program promotes advanced R&D for generating and nurturing breakthrough technologies and know-how (innovative), while also accelerating and deepening R&D that yields promising results.
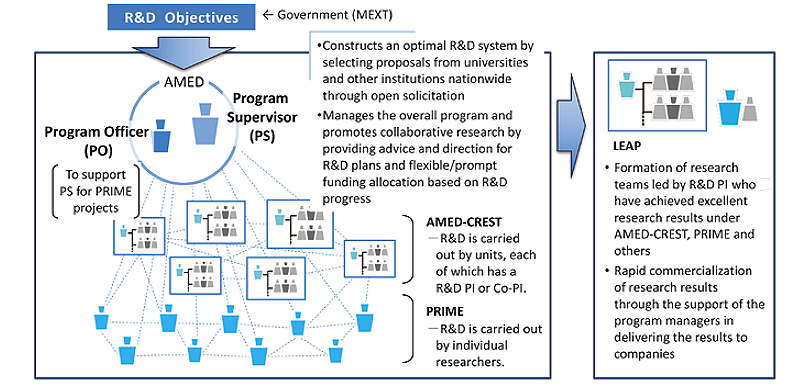
This program comprises three types of research: unit-type (AMED-CREST), solo-type (PRIME) and incubation-type (LEAP)*. For AMED-CREST and PRIME, AMED specifies the R&D pursuit areas and the Program Supervisors and Program Officers for leading the research under “Research and Development Objectives" designated by the national government. Through management by Program Supervisors and Program Officers and cooperation in each R&D area, the program aims to construct a R&D system transcending organizational frameworks as well as draw out the maximum potential of the research. AMED-CREST focuses on achieving world-class R&D results aimed at generating innovative seeds, with the respective R&D being conducted by a unit (a group of researchers) that is led by a R&D Principal Investigator (PI). PRIME aims to generate R&D results that will spawn innovative seeds, with the R&D being independently conducted by the individual R&D PI.
*: LEAP targets the swift commercialization of research results that are promising but where the risk of development is difficult for companies to evaluate. No solicitation is performed in LEAP.

Program Structure
In accordance with the “Plan for Promotion of Medical Research and Development", a Program Supervisor (hereinafter referred to as “PS") and Program Officer (hereinafter referred to as “PO") are assigned to the Program to ensure efficient utilization of competitive research funds and generation of excellent research accomplishments. The PS and PO have complete knowledge and understanding of the progress status of the program overall and provide the necessary guidance and advice to ensure that the program runs smoothly. Furthermore, research institutes and researchers are obligated to cooperate with the PS and PO. Based on the guidance and advice provided by the PS and PO, researchers may be required to revise, change, or suspend their R&D project plans or change their project implementation system if this is deemed necessary.
To accomplish the R&D Objectives designated by the national government (Ministry of Education, Culture, Sports, Science and Technology), the PS and PO construct a time-limited system for conducting R&D by organizing a R&D area, assembling an optimal mix of researchers from existing institutions and research projects -namely industry, academia and government- and oversee work in the R&D area with the cooperation of R&D Area Advisers and others. R&D PIs for AMED-CREST and PRIME receive support from the PS and PO in accordance with their operating policies as they advance the R&D projects they have proposed with the aim of generating innovative seeds in accordance with R&D Objectives and the management policies of the PS and PO, while actively building and utilizing personal networks through dialog with R&D Area Advisers and others, and coordination with participating researchers and connections with others both in Japan and overseas.
Research & Development Areas
AMED-CREST, PRIME
- Elucidation and Control of Biological Systems Leading to GENKI
- Understanding the mechanisms of sex and individual differences and advancing prediction technology
- Elucidation of mechanisms for stress responses to disease development
- Bridging the fundamental mechanism of aging and the effective treatment of age-related disease associated with impaired functional system
- Integrated understanding of multi-sensing networks and elucidation of their control mechanisms leading to the innovation of medical technologies
- Generating research infrastructure and novel technologies for anti-infective drug and vaccine discovery
- Understanding proteostasis and discovering innovative medical applications
- Understanding of the biological phenomena and responses at the early life stages to improve the quality of health and medical care
- Understanding of Pathophysiological Processes and Discovery of Medical Technology Seeds through Spatiotemporal Research of Tissue Adaptation and Repair Mechanisms
- Clarification of the Mechanisms of Individual’s Functional Impairment over the Entire Life Course
- Understanding the Interactions and Symbiosis between the Microbiome and the Host Organism, Leading to an Understanding of the Mechanisms of Disease Onset (Completed)
- Elucidation of Mechanobiological Mechanisms and Their Application to the Development of Innovative Medical Instruments and Technologies (Completed)
- Studies on Specific Activities and Functions of Lipid Molecules to Develop Innovative Medical Technologies (Completed)
- Creation of Innovative Technology for Medical Applications Based on the Global Analyses and Regulation of Disease-Related Metabolites(Completed)
- Innovation for Ideal Medical Treatment Based on the Understanding of Maintenance, Change and Breakdown Mechanisms of Homeostasis among Interacting Organ Systems(Completed)
- Development of Fundamental Technologies for Diagnosis and Therapy Based upon Epigenome Analysis (Completed)
- Creation of Basic Medical Technologies to Clarify and Control the Mechanisms Underlying Chronic Inflammation (Completed)
- Elucidation of the Principles of Formation and Function of the Brain Neural Network and Creation of Control Technologies (Completed)
- Fundamental Technologies for Medicine Concerning the Generation and Regulation of Induced Pluripotent Stem (iPS) Cells (Completed)
- Etiological Basics of and Techniques for Treatment of Allergic and Autoimmune Diseases (Completed)
FORCE
LEAP
Brochure
Last updated 12/12/25

