Objectives / Main projects covered by AMED
Objectives
Medical R&D in Japan was seen as suffering from the problems of lacking a system that provides sufficiently seamless funding, from basic research to practical application, and lacking a system that adequately implements clinical studies / trials, causing discovery of new drugs to take too long. In order to deal with these issues, the Japan Agency for Medical Research and Development aims to act as a 'control tower' that directs integrated research, from basic research to practical application. And since Japan is projected to become the world's first ultra-aging society, AMED aims to achieve the world's healthiest and longest-living people by creating the world's most advanced medical technologies and services, and also aims to become a pillar of Japan's economy by fostering medicine, drugs, and medical devices as strategic industries.


Promotion of R&D in the eight integrated projects defined in the 2nd Healthcare Policy and medium- to long-term plan
AMED has established eight “Integrated Projects” centered on modalities, and under the Program Director (PD) these will coordinate with the projects of related ministries and agencies, and will be centrally promoted from basic research to practical application. Research in specific disease areas will be promoted across the integrated projects so that it can be managed flexibly by the coordinators of each disease area.
AMED implements a technical approach with clearly defined development objectives such as “prevention, diagnosis, treatment, prognosis, and quality of life”, with a focus on extending healthy longevity.
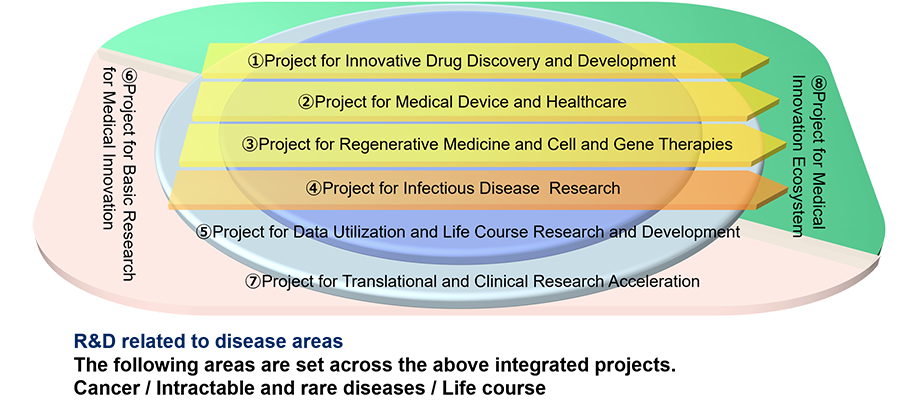
Main projects covered by AMED
Project for Innovative Drug Discovery and Development
Promotes a wide range of research and development, including drug discovery, clinical research as well as clinical trials. Also promote initiatives that contribute to the creation of innovative new drugs and the enhancement of drug discovery R&D capabilities.
Project for Medical Device and Healthcare
Promote research and development of medical devices and healthcare systems for advanced diagnosis and treatment, for which there is a great need in the medical field, and that contribute to prevention and improve the quality of life of the elderly.
Project for Regenerative Medicine and Cell and Gene Therapies
Discovering and fostering innovative seeds that could become new medical technologies in the fields of regenerative medicine, cell medicine, and gene therapy in which conducting research and development with a view to practical application and promoting clinical research and clinical trials.
Project for Infectious Disease Research
Promote research on a wide range of infectious diseases, including emerging and reemerging diseases, to gain new knowledge and develop preventive and therapeutic methods from normal times. In SCARDA, it promotes research that contributes to the development of domestic vaccines from basic to practical use.
Project for Data Utilization and Life Course Research and Development
Promote research and development that contributes to the prevention of disease onset and severity, clarification of pathological conditions, and diagnosis and treatment of diseases from a life-course perspective, utilizing disease registries for cancer, intractable diseases, dementia, etc.
Project for Basic Research for Medical Innovation
Stablish an R&D system that transcends the organizational and disciplinary boundaries of academia to promote basic research, including the creation and fostering of groundbreaking seeds for the creation of new modalities.
Project for Translational and Clinical Research Acceleration
Utilizing the functions of the Center for the Creation of Innovative Medical Technology, provide consistent support for bridging the gap from basic research to the clinical trial stage. Support the clinical research core hospitals to develop a system for conducting clinical trials at an international level.
Project for Medical Innovation Ecosystem
Strengthen R&D support for drug discovery ventures from the non-clinical to clinical trial stages and hands-on support by venture capitalists. Support for R&D conducted through collaboration between industry, academia, and government, and the development of a drug discovery ecosystem.
Last updated 05/22/25

